Green Hydrogen Generation through Direct Water Splitting
Hydrogen (H2) is considered a critical part of the global energy transition towards (net) zero carbon emission economy. H2 may substitute natural gas, especially in energy-thirsty industries, such as the steel and chemical industry resulting in a substantial decrease in its carbon (CO2) footprint. Furthermore, H2 may be used in the transportation sector fueling ships, trucks, and public transport. However, to effectively reduce the global CO2 footprint, green hydrogen is required, which is H2 generated by renewable energies – currently only about 0.1% of global H2 is green.
Challenges
To date, green H2 generation is very costly and inefficient and is mostly done by electrolysis powered by big solar power plants in regions with increased sun exposure.
To successfully develop green H2 into a future energy source its current technology needs to be improved substantially.
The direct generation of H2 from sunlight is a promising route for H2 generation. Combining solar cells with functional layers and catalysts enables water splitting by immersing the device in an aqueous medium. Electrolysis of water requires a voltage of 1,6 to 1,8V, hence single-junction solar cells are not suited as such voltage can only be achieved by tandem solar cells. Catalysts enable the oxidation of water at the anode and its reduction at the cathode, respectively leading to O2 and H2 evolution.
Since the device is placed in an electrolyte, anti-corrosive coatings are required to prevent the degradation of the semiconducting materials.
Choosing the right combination of absorber materials, catalysts and anti-corrosion coatings is the challenging part of designing such a sophisticated device.
III/V Semiconductor Tandem Solar Cell based Photocathode
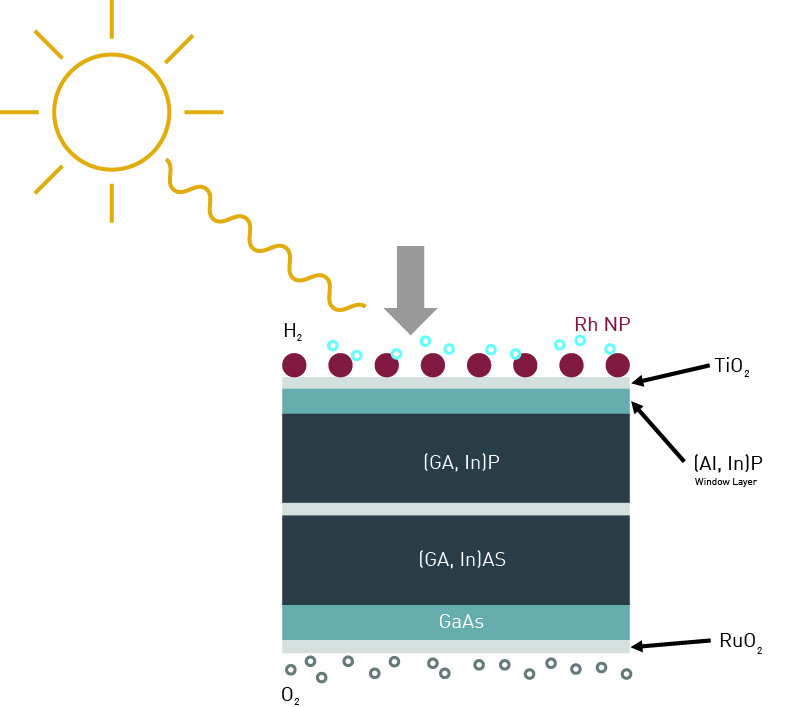
In 2018 an international team of researchers from the California Institute of Technology, the University of Cambridge, the Technical University Ilmenau, and the Fraunhofer Institute for Solar Energy Systems ISE published their work that showed an impressive 19% efficiency for their direct solar to hydrogen photochemical device1.
The device consists of a (Ga,In)P/(Ga,In)As tandem solar cell combined with dedicated functional layers enabling direct water splitting. The tandem solar cell is necessary to supply the required photovoltage for the electrolysis process. The TiO2 layer is deposited by ALD, using Titaniumtetraisopropoxide (TTIP) and H2O, and acts as anti-corrosion and antireflection coating. This is a critical step in enhancing the efficiency and lifetime of the device. Last, the cathode and anode are formed. For H2 generation Rhodium Nanoparticles with high surface area (Rh NPs), deposited by photoelectrodedeposition are used as catalysts, resulting in high exchange current and low light attenuation. The Ruthenium dioxide (RuO2) electrode for the oxidation reaction is deposited using a sputter process.
If you are interested in compound semiconductor solar cells have a look at our broad offer of CVD precursors for III/V semiconductors and suitable dopants. We also offer liquid Tertiarybutylphosphine (TBP) and Tertiarybutylarsine (TBAs) as proven alternatives to difficult to handle, highly toxic gaseous Phosphine and Arsine precursors.
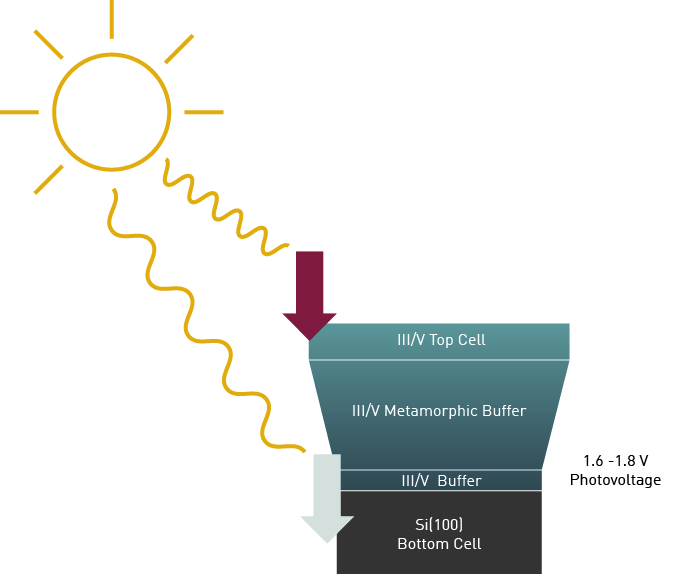
Tandem Solar Cell on Silicon based Photocathode
A slightly different approach would be to use conventional Silicon (Si) solar cells as bottom cell and integrate a III/V top cell above. The Si technology is already well known and Si substrates are cheap. The Fraunhofer Institute for Solar Energy Systems ISE is leading a consortium to develop such a device in the framework of the Project “H2Demo”.
Successful integration of III/V semiconductors on Si is challenging but can be achieved in a two-step process using the Precursors TBP and Triethylgallium (TEGa)2. In the first step, a GaP nucleation layer is deposited using flow rate modulated epitaxy at low temperatures that is subsequently overgrown by a GaP layer at higher temperatures in the second step.
To increase the lattice constant to the desired top cell material, Ga(As,P) is a suitable candidate, which requires an additional As precursor, such as Tertiarybutylarsine (TBAs).
References:
- Cheng, W.-H. et al. Monolithic Photoelectrochemical Device for Direct Water Splitting with 19% Efficiency. ACS Energy Lett 3, (2018) doi: 10.1021/acsenergylett.8b00920.
- Volz, K. et al. GaP-nucleation on exact Si (0 0 1) substrates for III/V device integration. J Cryst Growth 315, 37–47 (2011) doi: 10.1016/j.jcrysgro.2010.10.036.